How IV Pressure Infusion Bags Revolutionize Fluid Delivery in Critical Settings
- Share
- Issue Time
- Mar 25,2026
Summary
IV pressure infusion bags are crucial medical devices designed to facilitate rapid and controlled intravenous fluid and blood product administration, particularly in emergency and critical care settings. Unlike traditional gravity-fed systems, these bags apply external positive pressure, typically around 300 mmHg, using an inflatable cuff, a manual pump, and a manometer. This mechanism significantly accelerates fluid delivery, making them indispensable for managing conditions involving severe fl

As a healthcare professional, you understand the critical importance of rapid and precise fluid delivery, especially in emergency and critical care settings. While traditional gravity-fed IV systems have served us for years, they often fall short when speed is of the essence. This is where IV pressure infusion bags come into play, offering a revolutionary approach to patient care that can significantly impact patient outcomes.
Join us as we explore how these specialized medical devices work, why they are indispensable in various clinical scenarios, and the best practices for their safe and effective use. This comprehensive guide aims to provide a deep understanding of IV pressure infusion bags and affirm their vital role in modern medicine.
Key Takeaways
IV pressure infusion bags are essential tools in modern critical care, designed to overcome the limitations of traditional gravity-fed systems. Their primary function is to enable rapid and controlled administration of intravenous fluids, blood products, and medications by applying external, positive pressure to a standard IV bag. This capability is life-saving in situations marked by significant fluid loss or hemodynamic instability, such as trauma, major surgery, and septic shock. The core mechanism involves an inflatable cuff or bladder that encases the fluid container. A manual pump and a manometer allow clinicians to inflate the bladder to a specific pressure, typically around 300 mmHg, which forces the fluid out at a much higher rate than gravity alone could achieve.
The applications for these devices span multiple high-acuity environments. In emergency departments and trauma bays, they are fundamental for rapid fluid resuscitation in patients with hypovolemic shock. During complex surgical procedures, pressure infusors help anesthesiologists quickly replace blood loss and maintain stable blood pressure. In Intensive Care Units (ICUs), they are used not only for aggressive fluid management in conditions like sepsis but also for the crucial, albeit less dramatic, task of maintaining the patency of invasive arterial lines for continuous hemodynamic monitoring.
However, the power of these devices comes with significant responsibility. Safe and effective use hinges on strict adherence to established protocols and a vigilant awareness of potential complications. The most catastrophic risk is air embolism, which can occur if the system is not properly primed, if clamps are left closed while pressure is applied, or if a bag is allowed to run completely dry. To mitigate this, best practices include meticulous priming of all IV lines, confirming all clamps are open before infusion, and setting the "Volume to Be Infused" (VTBI) on smart pumps to a value slightly less than the total volume of the bag. Continuous monitoring of the patient for signs of fluid overload, regular checks of the equipment's pressure gauge, and understanding the specific mechanics of the infusion pumps being used—whether they are head-height dependent or use a cassette-based system—are all non-negotiable aspects of safe practice. Ultimately, mastery of IV pressure infusion bags requires a combination of robust technical knowledge, situational awareness, and an unwavering commitment to patient safety.
Understanding the Power of IV Pressure Infusion Bags: What They Are and How They Work
To truly appreciate the impact of IV pressure infusion bags, we must first go back to basics and understand the fundamental problem they solve. In clinical settings where every second counts, the slow, steady drip of a gravity-fed IV is often not enough. This section will define what these devices are and demystify the mechanics that make them so effective.
Defining IV Pressure Infusion Bags
At its core, an IV pressure infusion bag—also known as a pressure infusor or pressure bag—is a device designed to apply positive pressure to a flexible container of intravenous fluid. This external pressure forces the fluid to flow out of the bag and into the patient’s bloodstream at a much faster rate than would be possible with a standard gravity-drip infusion.
Beyond Gravity: The Need for Accelerated Fluid Delivery
Traditional IV setups rely solely on gravity. The rate of infusion is determined by the height of the IV bag relative to the patient and the resistance within the IV tubing. While reliable for routine hydration or medication delivery, this method has severe limitations in critical situations. In a patient suffering from hemorrhagic shock after a traumatic injury, for instance, replacing lost blood volume as quickly as possible is paramount to survival. A gravity infusion might deliver fluid at a rate of 200 mL per hour; a pressure infusion bag can escalate this to over 1,000 mL in a matter of minutes. This need for accelerated delivery is the primary driver behind the invention and widespread adoption of pressure infusion technology in medicine.
The Mechanics Behind IV Pressure Infusion Bags
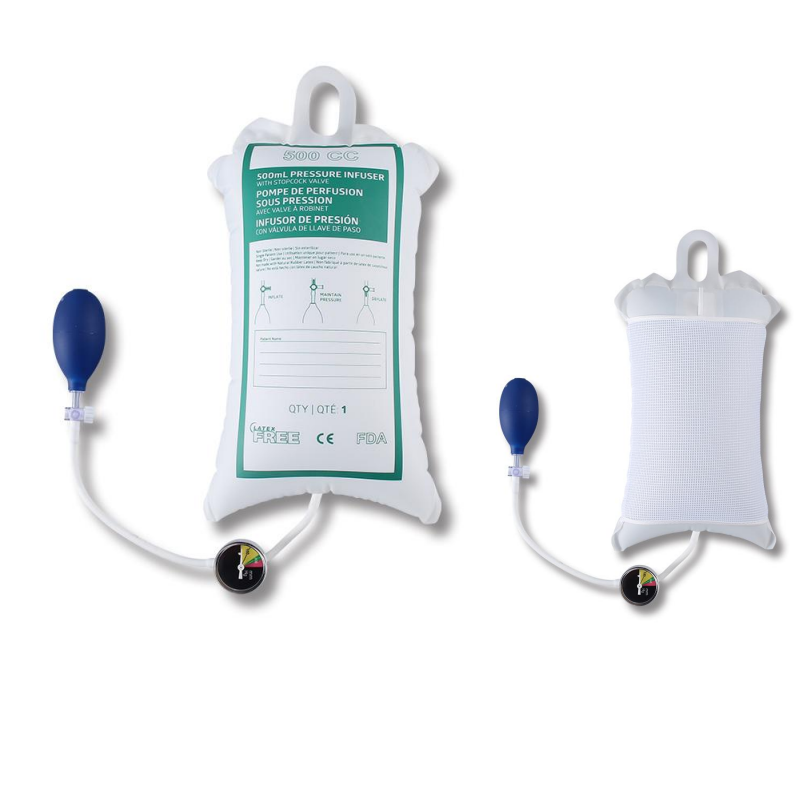
The design of a pressure infusion bag is elegantly simple yet highly effective. It consists of several key components working in concert to provide controlled, rapid infusion.
Key Components:
- Inflatable Bladder: A durable, cuff-like bag, typically made of nylon, that wraps around the IV fluid bag.
- Clear Sleeve: A transparent window or sleeve that holds the IV bag securely, allowing clinicians to monitor the fluid type and remaining volume.
- Hand Pump Bulb: A manual, squeezable bulb used to inflate the bladder with air.
- Pressure Gauge (Manometer): A color-coded analog or digital gauge that displays the amount of pressure within the bladder, usually measured in millimeters of mercury (mmHg). This is the most critical component for safe operation.
- Tubing and Valve: A system of tubes connects the bulb to the bladder, with a valve to control the inflation and deflation.
Inflation and Pressure Control: Achieving Optimal Flow
The process of using a pressure infusion bag is straightforward. The clinician places a bag of IV fluid or blood product inside the sleeve, hangs the entire device on an IV pole, and then inflates the outer bladder using the hand pump. As the bladder fills with air, it constricts and applies uniform pressure to the fluid bag inside.
The clinician carefully watches the pressure gauge, inflating the device to the target pressure, which is most commonly 300 mmHg. This pressure remains constant, ensuring a consistent and rapid flow rate until the bag is nearly empty. Maintaining this specific pressure is crucial; too little pressure will fail to achieve the desired flow, while excessive pressure could potentially damage the IV bag or, in rare cases, the device itself. This mechanism transforms a standard IV bag into a high-performance fluid delivery system, all without complex electronics.
The Indispensable Role of IV Pressure Infusion Bags in Critical Care
The true value of IV pressure infusion bags is most evident in the high-stakes environments of emergency medicine, surgery, and intensive care. In these settings, the ability to rapidly and precisely control fluid administration can be the difference between life and death.
Saving Lives: Rapid Fluid Resuscitation with IV Pressure Infusion Bags
Rapid fluid resuscitation is the cornerstone of treatment for various forms of shock, particularly hypovolemic shock resulting from massive fluid or blood loss. Pressure infusors are the primary tool for this intervention.
Emergency Departments and Trauma Bays: Time-Sensitive Interventions
In the chaotic environment of an emergency department (ED) or trauma bay, time is the most critical factor. A patient arriving with severe injuries from a car accident, a gunshot wound, or a major fall may be in hemorrhagic shock, a state where their body doesn't have enough blood volume to supply its organs with oxygen. Standard gravity infusions are far too slow to counteract this rapid loss.
By using a pressure infusion bag, the trauma team can infuse liters of crystalloid solution (like normal saline) or blood products in minutes, rapidly expanding the patient's blood volume to restore blood pressure and organ perfusion. This immediate intervention is a vital bridge that stabilizes the patient, allowing time for diagnostic imaging and definitive surgical treatment. Without the ability to deliver fluids at such high rates, many of these patients would not survive the initial "golden hour" of trauma care.
Precision and Control: Optimizing Blood Transfusions and Medications with IV Pressure Infusion Bags
Beyond sheer speed, pressure bags also offer enhanced control, which is essential for other critical care applications like blood transfusions and perioperative fluid management.
Surgical Settings: Managing Blood Loss and Anesthesia Support
During major surgical procedures, such as cardiovascular surgery, liver transplants, or complex orthopedic repairs, significant blood loss is an anticipated risk. The anesthesia team is responsible for maintaining the patient's hemodynamic stability throughout the operation. Pressure infusion bags are a standard part of their toolkit.
They allow for the rapid administration of packed red blood cells, plasma, and other fluids to precisely match the patient's losses in real-time. This not only prevents hypovolemia but also ensures that vital organs remain perfused under anesthesia. The ability to titrate fluid delivery so accurately helps prevent the complications of both under-resuscitation (shock, organ failure) and over-resuscitation (pulmonary edema, coagulopathy).
Intensive Care Units: Sustaining Critically Ill Patients
In the Intensive Care Unit (ICU), patients often require meticulous fluid management and invasive monitoring. IV pressure infusion bags serve two key roles in this setting. First, for patients in septic shock or other distributive shock states, large volumes of fluid are often needed quickly to support blood pressure. Pressure bags facilitate this initial, aggressive resuscitation phase.
Second, and just as important, is their role in maintaining arterial lines. These catheters, placed in an artery (usually the radial artery), provide beat-to-beat blood pressure monitoring and allow for frequent blood sampling. To keep the line from clotting, a small, continuous flow of saline is flushed through the system. This flush system is kept under constant pressure—typically 300 mmHg—using a dedicated pressure infusion bag. This ensures the accuracy of the blood pressure reading and the patency of the line, which is essential for managing critically ill patients on powerful medications.
Maximizing Safety and Efficiency with IV Pressure Infusion Bags
While IV pressure infusion bags are powerful tools, their misuse can lead to serious patient harm. A deep understanding of potential pitfalls and a strict adherence to best practices are absolutely essential for any clinician using this equipment. Safety cannot be an afterthought; it must be integrated into every step of the process.
Avoiding Common Pitfalls with IV Pressure Infusion Bags
Several common errors can turn this life-saving device into a potential hazard. Vigilance and a systematic approach are key to preventing these mistakes.
The Dangers of an Unopened Secondary Clamp
One of the most critical and easily avoidable errors is failing to open a clamp on the IV tubing before or immediately after applying pressure. If the roller clamp or a slide clamp on the IV administration set is left in the closed position while the bag is pressurized, the immense pressure (300 mmHg) has nowhere to go. This can lead to two dangerous outcomes: the IV bag could rupture, spilling its contents and delaying care, or, far more dangerously, if the fluid runs out, the system could drive a massive volume of air into the line once the clamp is finally opened, risking a catastrophic air embolism.
Accurate Volume to Be Infused (VTBI) Programming for IV Pressure Infusion Bags
When using a pressure bag in conjunction with a smart infusion pump, another layer of safety is available. Clinicians must program the pump's "Volume to Be Infused" (VTBI) to be slightly less than the total volume in the IV bag (e.g., setting the VTBI to 950 mL for a 1000 mL bag). This ensures that the pump will alarm and stop the infusion before the bag runs completely dry. Neglecting this step defeats a key safety feature of the pump, creating a scenario where the pump could continue to operate on an empty bag, pushing air down the line under high pressure.
Preventing Complications: Addressing Check Valve Failure and Simultaneous Flow Issues with IV Pressure Infusion Bags
Many patients in critical care receive multiple IV infusions simultaneously through a single IV access site. These setups rely on one-way check valves in the IV tubing to prevent fluid from one line from flowing backward into another. If a pressure infusion bag is used on a primary infusion, it creates a high-pressure gradient. If the check valve on a secondary line (e.g., a medication drip) fails, the high-pressure fluid from the primary line can flow backward into the lower-pressure secondary line. This can lead to under-delivery or non-delivery of the critical secondary medication and cause the medication to mix incorrectly in the tubing, posing a significant risk to the patient. Regular inspection of equipment and understanding the dynamics of simultaneous infusions are crucial.
Best Practices for Using IV Pressure Infusion Bags
Adhering to a standardized set of best practices can significantly mitigate the risks associated with pressure infusions and ensure patient safety.
Adhering to Manufacturer Guidelines for IV Pressure Infusion Bags
Not all pressure bags are created equal. They come in reusable and disposable models, with different pressure limits and cleaning requirements. It is imperative to always follow the specific manufacturer's Instructions for Use (IFU). This includes:
- Pressure Limits: Never inflate a bag beyond its specified limit, which is almost universally 300 mmHg.
- Cleaning and Inspection: Reusable bags must be properly cleaned and inspected for wear and tear, rips, or faulty gauges before each use. A compromised bag may not hold pressure or could fail during use.
- Compatibility: Ensure the pressure bag is appropriately sized for the fluid container being used.
Continuous Monitoring and Vigilance with IV Pressure Infusion Bags
Using a pressure infusion bag is not a "set it and forget it" procedure. It demands continuous vigilance from the healthcare team.
- Patient Monitoring: Closely monitor the patient's vital signs (blood pressure, heart rate, respiratory rate, oxygen saturation) and watch for signs of fluid overload, such as new or worsening crackles in the lungs or swelling (edema).
- Site Monitoring: Regularly check the IV insertion site for signs of infiltration or extravasation (fluid leaking into the surrounding tissue). Under high pressure, an infiltration can escalate quickly, causing significant tissue damage.
- Equipment Monitoring: Keep a close eye on the pressure gauge to ensure it remains at the target level and frequently check the remaining fluid volume in the bag to anticipate when a new bag will be needed and prevent it from running dry.
| Best Practice | Rationale |
|---|---|
| Verify All Clamps Are Open | Prevents bag rupture and the risk of air embolism from a pressurized, empty system. |
| Set VTBI Less Than Bag Volume | Uses the smart pump's safety feature to stop infusion before the bag is empty, preventing air entrainment. |
| Use on a Dedicated Line if Possible | Minimizes the risk of backflow into secondary infusions if a one-way valve fails. |
| Follow Manufacturer IFU | Ensures correct pressure limits, cleaning procedures, and operational use for the specific device model. |
| Continuous Monitoring | Allows for immediate detection of patient complications (fluid overload), IV site issues (infiltration), and equipment problems. |
Comparing IV Pressure Infusion Bags: Head-Height Dependent vs. Cassette IV Smart Pump Systems
The effectiveness and safety of a pressure infusion are not solely dependent on the pressure bag itself but also on the type of intravenous smart pump it's used with. The two main categories of infusion pumps—head-height dependent systems and cassette-based systems—interact with pressure bags in fundamentally different ways. Understanding these differences is crucial for optimizing clinical practice.
The Head-Height Differential Method for IV Pressure Infusion Bags
Many of the most common IV pumps on the market are head-height dependent. This means their mechanism relies on a certain amount of gravity assistance, and their accuracy can be affected by the height of the IV bag in relation to the pump's internal pumping chamber.
Setup Requirements and Considerations
In a standard gravity infusion, raising the IV bag higher on the pole increases the "head height," which increases the flow rate. Head-height dependent pumps are engineered with this principle in mind. However, when you introduce a pressure infusion bag, you are essentially overriding the gravity component with 300 mmHg of external force. While this achieves the goal of rapid infusion, it can create inconsistencies if the pump's software isn't fully designed to account for such a high inlet pressure. Clinicians must be aware that while the pump may still be counting volume, the actual flow dynamics are being dictated by the pressure bag, not the pump's peristaltic mechanism alone.
The Cassette Method for IV Pressure Infusion Bags
An alternative technology is the cassette-based infusion pump. These pumps utilize a dedicated, disposable cassette that contains the pumping mechanism and is not reliant on the head height of the fluid container.
Simplified Setup and Enhanced Safety Features
In a cassette system, the pump's mechanism directly acts on the fluid within the cassette, isolating the pumping action from the pressure in the line leading from the IV bag. This design offers several advantages when used with a pressure infusion bag. The flow rate is determined more precisely by the pump's programming because it is not influenced by the variable head height or the external pressure from the bag. This can lead to a more predictable and controlled infusion, even at high rates. Furthermore, some cassette systems have enhanced features to detect air-in-line more effectively, providing an additional layer of safety during high-risk pressure infusions.
Why Understanding the Differences in IV Pressure Infusion Bags Matters
The choice between a head-height dependent system and a cassette-based system has real-world implications for patient safety and clinical workflow. A clinician who is accustomed to the feedback and behavior of a cassette pump may be surprised by the nuances of a head-height dependent pump when a pressure bag is applied, and vice versa.
Understanding the mechanics of the specific equipment available in your institution is paramount. It influences:
- Flow Rate Accuracy: Cassette systems may offer more reliable accuracy during pressure infusions.
- Alarm Response: The types and sensitivity of alarms (e.g., occlusion, air-in-line) can differ significantly.
- Setup Complexity: While both are straightforward, the underlying mechanics can affect troubleshooting when problems arise.
Ultimately, regardless of the pump type, the fundamental principles of safe pressure infusion remain the same: meticulous priming, clamp management, VTBI setting, and constant monitoring. However, knowing the technology you are working with allows for a higher level of mastery and safer patient care.
Frequently Asked Questions (FAQs) About IV Pressure Infusion Bags
1. What is the maximum recommended pressure for an IV pressure infusion bag?
The almost universal standard and maximum recommended pressure for medical pressure infusion bags is 300 mmHg. This pressure is recognized as providing a safe and effective balance for rapid infusion without posing an undue risk to the integrity of the IV fluid container or the device itself. Most pressure gauges are color-coded, with a green zone that indicates the operational pressure up to 300 mmHg and a red zone beyond that to warn against over-inflation. It is critical to never exceed the manufacturer's specified limit, which is consistently 300 mmHg for all major models used in clinical practice.
2. Can you use a pressure infusion bag with any type of IV fluid or blood product?
Generally, yes. Pressure infusion bags are compatible with standard crystalloid solutions (e.g., Normal Saline, Lactated Ringer's) and colloid solutions. They are also routinely and safely used for the rapid transfusion of blood products like packed red blood cells (PRBCs), fresh frozen plasma (FFP), and platelets. However, some historical concern existed regarding potential damage (hemolysis) to red blood cells under pressure. Modern studies have largely shown that infusion at 300 mmHg does not cause clinically significant hemolysis. That said, it is always best practice to use the largest-bore IV catheter possible for blood transfusions to minimize shear stress on the cells as they enter the vein.
3. What is the single biggest risk of using a pressure infusion bag, and how is it prevented?
The single most feared and catastrophic complication of using an IV pressure infusion bag is a massive air embolism. This occurs when a significant volume of air enters the venous system and travels to the heart and lungs, causing cardiovascular collapse. This can happen if an empty or partially empty IV bag is pressurized and the air is forced down the tubing into the patient. Prevention is the only effective treatment and relies on a multi-layered, vigilant approach:
- Meticulous Priming: Ensure every millimeter of the IV tubing is completely filled with fluid and all air bubbles are removed before connecting to the patient.
- Never Let the Bag Run Dry: Constant monitoring of the fluid level is essential.
- Use Smart Pump Safeguards: Always program the VTBI (Volume To Be Infused) on the infusion pump to be 50-100 mL less than the total bag volume.
- Constant Vigilance: Never leave a pressure infusion unattended for an extended period.
4. Are IV pressure infusion bags reusable or single-use?
This depends entirely on the specific model. The market includes both reusable and disposable options.
- Reusable Bags: These are typically made of durable nylon and are designed to be cleaned and disinfected between patients according to manufacturer and institutional protocols. They represent a higher initial cost but can be more economical over time. They require a rigorous process for cleaning, inspection (for tears, leaks, or gauge malfunction), and storage.
- Disposable Bags: These single-use models are designed to be discarded after use on a single patient. They eliminate the risks associated with cross-contamination and the labor involved in cleaning, but they generate more medical waste and have a higher per-use cost. Many institutions are moving towards disposable models to simplify infection control.
5. Besides trauma, what is another common, critical use for a pressure infusion bag?
One of the most common and vital uses for a pressure infusion bag outside of rapid volume resuscitation is for the maintenance of an invasive arterial line monitoring system. An arterial line, which provides continuous, real-time blood pressure measurement, must be connected to a transducer. This entire system is kept patent and functional by a continuous, slow flush of saline. To ensure the saline flush system works correctly and to prevent blood from backing up into the tubing from arterial pressure, the saline bag is placed in a pressure infusion bag and maintained at a constant pressure of 300 mmHg. This ensures the accuracy of the hemodynamic data, which is crucial for making treatment decisions for critically ill patients in ICUs and operating rooms.
Conclusion
IV pressure infusion bags are far more than just supplemental medical devices; they are indispensable, life-saving tools that empower healthcare professionals to deliver rapid, precise, and controlled fluid therapy in the most critical of situations. From the trauma bay to the operating room to the intensive care unit, their ability to quickly reverse hypovolemia and stabilize patients is unparalleled. By truly understanding their mechanics, diverse applications, and the vital best practices for their use, we can continue to revolutionize fluid delivery and secure significantly improved patient outcomes.
I am confident that a commitment to continuous education and the strict application of safety protocols in the use of IV pressure infusion bags is essential for optimizing patient safety and elevating the standard of care across all acute and critical care environments. We encourage you to share this article with your colleagues and contribute to a culture of safety and excellence. The power of these devices is matched only by the skill and diligence of the clinicians who wield them.